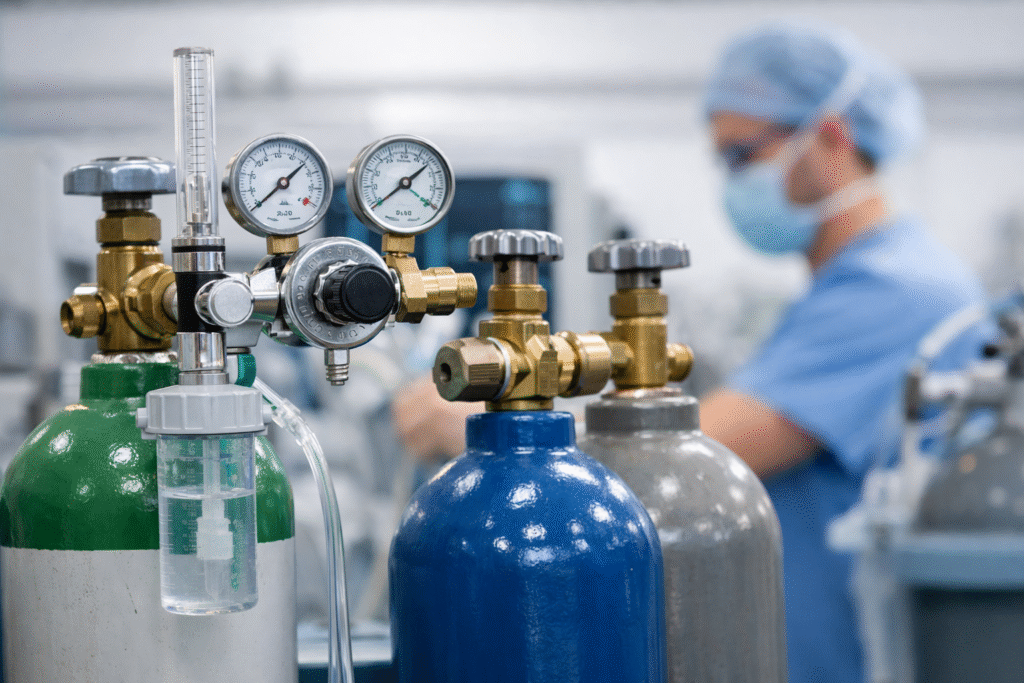
What manufacturers, importers, and distributors need to know about the new FDA certification framework now in effect.
On February 12, 2026, the U.S. Food and Drug Administration (FDA) released revised draft guidance for industry titled Certification Process for Designated Medical Gases. The guidance reflects regulatory changes stemming from the FDA’s final rule issued on June 18, 2024, which implemented Section 576 of the Federal Food, Drug, and Cosmetic Act (FD&C Act). Those regulations officially became effective on December 18, 2025.
The updated guidance provides clarity on how FDA administers the certification process for designated medical gases and what manufacturers must submit to demonstrate compliance.
What Are Designated Medical Gases?
Designated medical gases are specific gases intended for medical use that meet established compendial standards. The FDA recognizes seven designated medical gases:
- Oxygen
- Nitrogen
- Nitrous oxide
- Carbon dioxide
- Helium
- Medical air
- Carbon monoxide
These gases are widely used in healthcare settings for respiratory support, anesthesia, diagnostic procedures, and other therapeutic purposes.
Who Must Submit a Certification Request?
Manufacturers and certain distributors of designated medical gases must submit certification requests to the FDA to legally market their products under the new regulatory framework.
The guidance clarifies:
- Which entities are responsible for submitting certification requests
- When certification is required
- How imported medical gases are treated under the regulations
- The role of contract manufacturers and repackagers
For companies involved in cross-border supply chains, this certification requirement may directly affect import compliance, documentation, and regulatory filings.
What Information Must Be Included in a Certification Submission?
The revised guidance explains the required content for certification requests, including:
- Product identification and composition
- Manufacturing processes and controls
- Evidence demonstrating compliance with compendial standards
- Labeling information
- Facility details
FDA also outlines how it evaluates submissions and the potential outcomes, including certification approval, requests for additional information, or denial.
Why This Update Matters for the Supply Chain
The updated certification framework introduces clearer regulatory expectations but also increases compliance responsibilities for manufacturers and importers.
Potential impacts include:
- Additional documentation requirements for imports
- Changes to product labeling and registration processes
- Compliance reviews during customs clearance
- Increased regulatory scrutiny for medical-grade gas supply chains
Companies importing medical gases or related products should ensure their suppliers are properly certified and that documentation aligns with FDA requirements to avoid delays or enforcement actions.
How Alba Can Help
Regulatory changes affecting healthcare products can create uncertainty across global supply chains. Alba’s compliance specialists monitor FDA developments and help clients navigate evolving import requirements, documentation standards, and regulatory risks.
If your organization imports or distributes medical gases, now is the time to review supplier certifications and compliance procedures.
Have questions? Ask Alba.


